- Implement & Learn
Implementation Products - Resources
- For Consultants
- About Us
ISO 13485 Expert - Contact Us
You really can implement ISO 13485 and MDR by yourself – all you need is our documentation toolkits, along with included guidance and support. Our toolkits and other resources were developed for ease of use and to be understandable, with no expert knowledge required.
What’s more, here at the 13485Academy, we eliminate the stress and headache that can come with doing it yourself, and you’ll eliminate the huge price tag that comes with a consultant.
We’ve calculated your savings, and we estimate that you will save over 90% compared to the cost of a consultant. In other words, you’ll save thousands of dollars – with no drop in quality!
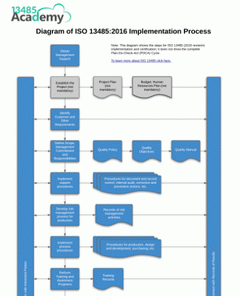
Planning the implementation of ISO 13485:2016 is a crucial step in the success of your Medical Device Management System. With our ISO 13485:2016 Implementation Diagram you can see at a glance the step-by-step process to follow, ensuring nothing is forgotten.

Knowing what documents and records are necessary for ISO 13485:2016 can be confusing. This white paper is designed to clear up any misunderstandings regarding documents required by this standard, as well as outlining non-mandatory documents that are commonly used. It also offers help on structuring these documents, in a straight-forward and easy-to-follow format.
This course teaches the core ISO 13485 standard, including all of the best practices for compliance.
See details...This course teaches you all that you need to know to perform an internal audit of the ISO 13485 standard for your company.
See details...This course teaches you the core principles you need to become an auditor for the ISO 13485 standard and offers accredited certification from ASIC.
See details...This course teaches all you need to know about ISO 13485, helping you become an independent consultant for the standard, and offers accredited certification from ASIC.
See details...

Our toolkits are developed by
ISO 13485 experts.

You’ll save time, money, and effort in your
ISO 13485 implementation.

We aren’t just a website – we’re real people
here to help you.