Mark Hammar
Mark Hammar
January 21, 2015
Implementation, maintenance, training, and knowledge products for Information Security Management Systems (ISMS) according to the ISO 27001 standard.
Automate your ISMS implementation and maintenance with the Risk Register, Statement of Applicability, and wizards for all required documents.
All required policies, procedures, and forms to implement an ISMS according to ISO 27001.
Train your key people about ISO 27001 requirements and provide cybersecurity awareness training to all of your employees.
Accredited courses for individuals and security professionals who want the highest-quality training and certification.
Create ISO 27001 documentation, get instant answers to any questions related to ISO 27001 and the ISMS, refine your writing, and build security training materials faster with Advisera’s AI-powered platform.
Compliance and training products for critical infrastructure organizations for the European Union’s Network and Information Systems cybersecurity directive.
All required policies, procedures, and forms to comply with the NIS 2 cybersecurity directive.
Company-wide training program for employees and senior management to comply with Article 20 of the NIS 2 cybersecurity directive.
Create NIS2 documentation, refine your writing, and build security awareness training materials faster with Advisera’s AI-powered platform.
Compliance and training products for financial entities for the European Union’s DORA regulation.
All required policies, procedures, and forms to comply with the DORA regulation.
Company-wide cybersecurity and resilience training program for all employees, to train them and raise awareness about ICT risk management.
Accredited courses for individuals and DORA professionals who want the highest-quality training and certification.
Create DORA documentation, refine your writing, and build financial sector cybersecurity training materials faster with Advisera’s AI-powered platform.
Training products for Artificial Intelligence Management Systems (AIMS) and AI governance according to the ISO 42001 standard.
Accredited courses for individuals, consultants, and AI professionals who want the highest-quality training and certification in AI governance and compliance.
Train your key people on ISO 42001 requirements and provide company-wide AI governance training so employees learn how to use AI responsibly and in compliance with your policies.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Compliance and training products for personal data protection according to the European Union’s General Data Protection Regulation.
All required policies, procedures, and forms to comply with the EU GDPR privacy regulation.
Train your key people about GDPR requirements to ensure awareness of data protection principles, privacy rights, and regulatory compliance.
Accredited courses for individuals and privacy professionals who want the highest-quality training and certification.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation, training, and knowledge products for Quality Management Systems (QMS) according to the ISO 9001 standard.
All required policies, procedures, and forms to implement a QMS according to ISO 9001.
Accredited courses for individuals and quality professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 and the QMS, refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for Environmental Management Systems (EMS) according to the ISO 14001 standard.
All required policies, procedures, and forms to implement an EMS according to ISO 14001.
Accredited courses for individuals and environmental professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 14001 and the EMS, refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation and training products for Occupational Health & Safety Management Systems (OHSMS) according to the ISO 45001 standard.
All required policies, procedures, and forms to implement an OHSMS according to ISO 45001.
Accredited courses for individuals and health & safety professionals who want the highest-quality training and certification.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation and training products for medical device Quality Management Systems (QMS) according to the ISO 13485 standard.
All required policies, procedures, and forms to implement a medical device QMS according to ISO 13485.
Accredited courses for individuals and medical device professionals who want the highest-quality training and certification.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Compliance products for the European Union’s Medical Device Regulation.
All required policies, procedures, and forms to comply with the EU MDR.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation products for Information Technology Service Management Systems (ITSMS) according to the ISO 20000 standard.
All required policies, procedures, and forms to implement an ITSMS according to ISO 20000.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation products for Business Continuity Management Systems (BCMS) according to the ISO 22301 standard.
All required policies, procedures, and forms to implement a BCMS according to ISO 22301.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation products for testing and calibration laboratories according to the ISO 17025 standard.
All required policies, procedures, and forms to implement ISO 17025 in a laboratory.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation products for automotive Quality Management Systems (QMS) according to the IATF 16949 standard.
All required policies, procedures, and forms to implement an automotive QMS according to IATF 16949.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation products for aerospace Quality Management Systems (QMS) according to the AS9100 standard.
All required policies, procedures, and forms to implement an aerospace QMS according to AS9100.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation, maintenance, training, and knowledge products for consultancies.
Handle multiple ISO 27001 projects by automating repetitive tasks during ISMS implementation.
All required policies, procedures, and forms to implement various standards and regulations for your clients.
Grow your business by organizing cybersecurity and compliance training for your clients under your own brand using Advisera’s learning management system platform.
Accredited Lead Auditor and Implementer courses for DORA and ISO standards, and advanced courses to help consultants grow their business, increase recurring revenue, and stand out from bigger competitors.
Create compliance documents, get instant answers to compliance questions, build training materials faster, and refine writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Find new clients, potential partners, and collaborators and meet a community of like-minded professionals locally and globally.
Implementation, maintenance, training, and knowledge products for the IT industry.
Automate your ISMS implementation and maintenance with the Risk Register, Statement of Applicability, and wizards for all required documents.
Documentation to comply with ISO 27001 (cybersecurity), ISO 22301 (business continuity), ISO 20000 (IT service management), GDPR (privacy), NIS 2 (critical infrastructure cybersecurity), and DORA (cybersecurity for financial sector).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity, privacy, and AI program.
Accredited courses for individuals and security professionals who want the highest-quality training and certification.
Create ISO 27001 and NIS2 documentation, get instant answers to any questions related to ISO 27001 and the ISMS, refine your writing, and build security training materials faster with Advisera’s AI-powered platform
Compliance, training, and knowledge products for essential and important organizations.
Documentation to comply with NIS 2 (cybersecurity), GDPR (privacy), ISO 27001 (cybersecurity), and ISO 22301 (business continuity).
Company-wide training program for employees and senior management to comply with Article 20 of the NIS 2 cybersecurity directive.
Accredited courses for individuals and security professionals who want the highest-quality training and certification.
Create ISO 27001 and NIS2 documentation, get instant answers to any questions related to ISO 27001 (ISMS), refine your writing, and build security awareness training materials faster with Advisera’s AI-powered platform
Implementation, training, and knowledge products for manufacturing companies.
Documentation to comply with ISO 9001 (quality), ISO 14001 (environmental), and ISO 45001 (health & safety), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS) and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for transportation & distribution companies.
Documentation to comply with ISO 9001 (quality), ISO 14001 (environmental), and ISO 45001 (health & safety), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS) and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for schools, universities, and other educational organizations.
Documentation to comply with ISO 27001 (cybersecurity), ISO 9001 (quality), and GDPR (privacy).
Company-wide cybersecurity and AI governance awareness program for all employees, to decrease incidents, support a successful cybersecurity program, and ensure responsible use of AI.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS) and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, maintenance, training, and knowledge products for telecoms.
Automate your ISMS implementation and maintenance with the Risk Register, Statement of Applicability, and wizards for all required documents.
Documentation to comply with ISO 27001 (cybersecurity), ISO 22301 (business continuity), ISO 20000 (IT service management), GDPR (privacy), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity program.
Accredited courses for individuals and security professionals who want the highest-quality training and certification.
Create ISO 27001 and NIS2 documentation, get instant answers to any questions related to ISO 27001 (ISMS), refine your writing, and build security awareness training materials faster with Advisera’s AI-powered platform.
Implementation, maintenance, training, and knowledge products for banks, insurance companies, and other financial organizations.
Automate your ISMS implementation and maintenance with the Risk Register, Statement of Applicability, and wizards for all required documents.
Documentation to comply with DORA (cybersecurity for financial sector), ISO 27001 (cybersecurity), ISO 22301 (business continuity), and GDPR (privacy).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity, privacy, and AI program.
Accredited courses for individuals and security professionals who want the highest-quality training and certification.
Create ISO 27001 and NIS2 documentation, get instant answers to any questions related to ISO 27001 (ISMS), refine your writing, and build security awareness training materials faster with Advisera’s AI-powered platform.
Implementation, training, and knowledge products for local, regional, and national government entities.
Documentation to comply with ISO 27001 (cybersecurity), ISO 9001 (quality), GDPR (privacy), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity, privacy, and AI program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 27001 (ISMS) and ISO 9001 (QMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for hospitals and other health organizations.
Documentation to comply with ISO 27001 (cybersecurity), ISO 9001 (quality), ISO 14001 (environmental), ISO 45001 (health & safety), NIS 2 (critical infrastructure cybersecurity) and GDPR (privacy).
Company-wide cybersecurity and AI governance awareness program for all employees, to decrease incidents, support a successful cybersecurity program, and ensure responsible use of AI.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 27001 (ISMS), ISO 9001 (QMS), and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for the medical device industry.
Documentation to comply with ISO 13485 (medical device), ISO 27001 (cybersecurity), ISO 9001 (quality), ISO 14001 (environmental), ISO 45001 (health & safety), NIS 2 (critical infrastructure cybersecurity) and GDPR (privacy).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity, privacy, and AI program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 27001 (ISMS), ISO 9001 (QMS), and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for the aerospace industry.
Documentation to comply with AS9100 (aerospace), ISO 9001 (quality), ISO 14001 (environmental), and ISO 45001 (health & safety), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS) and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for the automotive industry.
Documentation to comply with IATF 16949 (automotive), ISO 9001 (quality), ISO 14001 (environmental), and ISO 45001 (health & safety), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity and AI program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS) and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for laboratories.
Documentation to comply with ISO 17025 (testing and calibration laboratories), ISO 9001 (quality), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity program.
Accredited courses for individuals and quality professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
 Mark Hammar
Mark Hammar
Note: This article was updated according to the ISO 13485:2016 revision.
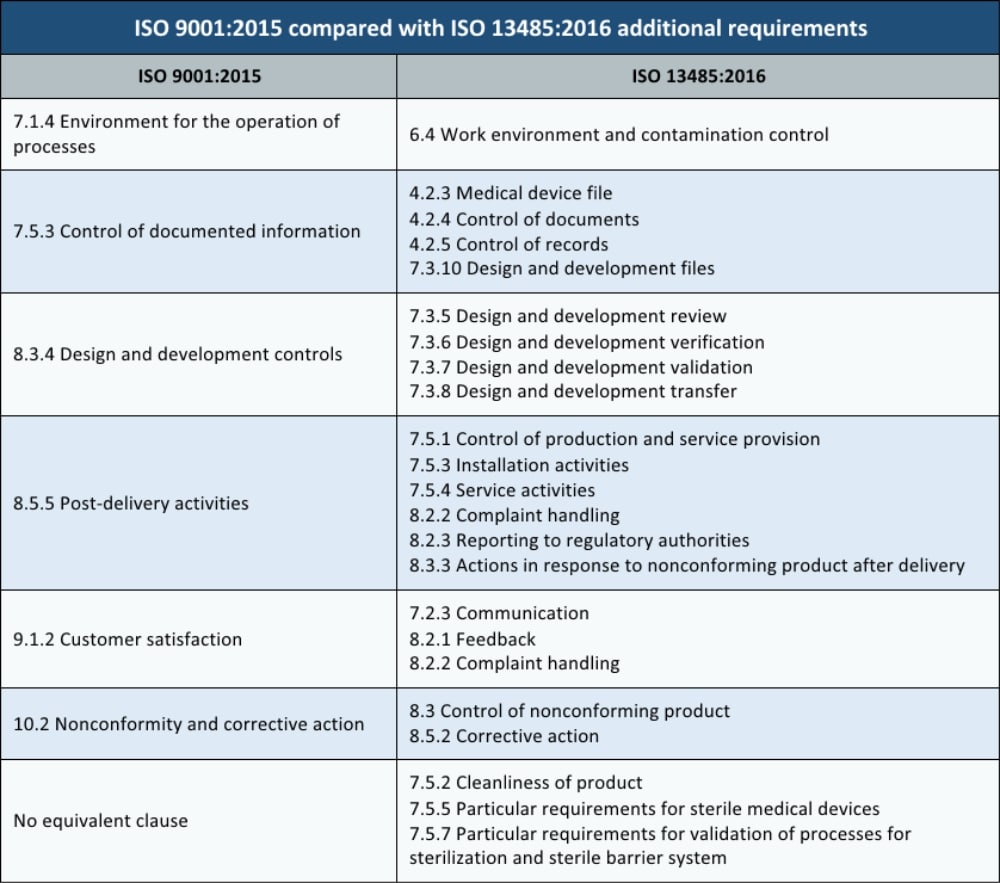
ISO 13485 is the international standard requirement for a medical device quality management system. Like many other quality management system requirements for special purposes (such as IATF 16949 for automotive production and service parts and AS9100 for use by aviation, space and defense organizations), the ISO 13485 standard is based on the requirements of ISO 9001.
Just like these other standards, ISO 13485 includes the entire ISO 9001 standard with additional requirements included in blue italics text. One major distinction of ISO 13485 is that it is intended to also be required for regulatory purposes as well as a non-statutory requirement for a quality management system.
Until the introduction of the high-level structure in 2015, ISO 9001 and ISO 13485 were very similar standards and it was very easy to build a quality management system that met the requirements of both of them. However, today the situation is slightly different because ISO 13485:2016 still supports the structure of old ISO 9001 revision (from 2008).
The question everyone is asking is why 13485:2016 is so different from 9001:2015, when 13485:2016 came out just 6 months after 9001:2015 was released. Unfortunately, corrections on the ISO 13485:2003 took a long time. The new version of ISO 13485 was ready to be released in 2016 although it relied on ISO 9001:2008. So, when ISO 9001:2015 was finally released with the new structure, 13485:2016 was also already ready for release with the ISO 9001:2008 structure. The ISO organization decided to release 13485 with the old structure.
For information on the differences between ISO 9001:2015 and ISO 9001:2008, see Infographic: ISO 9001:2015 vs. 2008 revision – What has changed?
Besides these differences in the structure, there are also similarities between ISO 9001:2015 and ISO 13485:2016:
In ISO 13485:2016, the manufacturer must show compliance with regulatory requirements in 58 places. For comparison, in ISO 9001:2015 the term ‘’regulatory requirements’’ is only mentioned 11 times.
After some information added in the introductory section that mainly tailors the text to the medical device industry, the first inclusions are in the terms and definitions. There are 14 new terms used by the medical device industry. The differences between manufacturers, importers, distributors, and terms such as clinical evaluation and post-market surveillance are specifically explained.
Listed below are additional requirements for ISO 13485:2016 when compared to ISO 9001:2015:
Clause 4 – Quality Management System
Clause 5 – Management Responsibility
Clause 6 – Resource Management
Clause 7 – Product Realization
Clause 8 – Measurement, Analysis and Improvement
If your organization is at all involved in the medical device industry, ISO 13485 is the QMS standard you should look at for additional requirements above and beyond ISO 9001.
To implement ISO 13485 easily and efficiently, use our ISO 13485 Documentation Toolkit that provides step-by-step guidance and all documents for full ISO 13485 compliance.

Mark Hammar is a Certified Manager of Quality / Organizational Excellence through the American Society for Quality and has been a Quality Professional since 1994. Mark has experience in auditing, improving processes, and writing procedures for Quality, Environmental, and Occupational Health & Safety Management Systems, and is certified as a Lead Auditor for ISO 9001, AS9100, and ISO 14001.

You may unsubscribe at any time. For more information, please see our privacy notice.