 Mark Hammar
Mark Hammar
March 10, 2015
Implementation, maintenance, training, and knowledge products for Information Security Management Systems (ISMS) according to the ISO 27001 standard.
Automate your ISMS implementation and maintenance with the Risk Register, Statement of Applicability, and wizards for all required documents.
All required policies, procedures, and forms to implement an ISMS according to ISO 27001.
Train your key people about ISO 27001 requirements and provide cybersecurity awareness training to all of your employees.
Accredited courses for individuals and security professionals who want the highest-quality training and certification.
Create ISO 27001 documentation, get instant answers to any questions related to ISO 27001 and the ISMS, refine your writing, and build security training materials faster with Advisera’s AI-powered platform.
Compliance and training products for critical infrastructure organizations for the European Union’s Network and Information Systems cybersecurity directive.
All required policies, procedures, and forms to comply with the NIS 2 cybersecurity directive.
Company-wide training program for employees and senior management to comply with Article 20 of the NIS 2 cybersecurity directive.
Create NIS2 documentation, refine your writing, and build security awareness training materials faster with Advisera’s AI-powered platform.
Compliance and training products for financial entities for the European Union’s DORA regulation.
All required policies, procedures, and forms to comply with the DORA regulation.
Company-wide cybersecurity and resilience training program for all employees, to train them and raise awareness about ICT risk management.
Accredited courses for individuals and DORA professionals who want the highest-quality training and certification.
Create DORA documentation, refine your writing, and build financial sector cybersecurity training materials faster with Advisera’s AI-powered platform.
Training products for Artificial Intelligence Management Systems (AIMS) and AI governance according to the ISO 42001 standard.
Accredited courses for individuals, consultants, and AI professionals who want the highest-quality training and certification in AI governance and compliance.
Train your key people on ISO 42001 requirements and provide company-wide AI governance training so employees learn how to use AI responsibly and in compliance with your policies.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Compliance and training products for personal data protection according to the European Union’s General Data Protection Regulation.
All required policies, procedures, and forms to comply with the EU GDPR privacy regulation.
Train your key people about GDPR requirements to ensure awareness of data protection principles, privacy rights, and regulatory compliance.
Accredited courses for individuals and privacy professionals who want the highest-quality training and certification.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation, training, and knowledge products for Quality Management Systems (QMS) according to the ISO 9001 standard.
All required policies, procedures, and forms to implement a QMS according to ISO 9001.
Accredited courses for individuals and quality professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 and the QMS, refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for Environmental Management Systems (EMS) according to the ISO 14001 standard.
All required policies, procedures, and forms to implement an EMS according to ISO 14001.
Accredited courses for individuals and environmental professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 14001 and the EMS, refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation and training products for Occupational Health & Safety Management Systems (OHSMS) according to the ISO 45001 standard.
All required policies, procedures, and forms to implement an OHSMS according to ISO 45001.
Accredited courses for individuals and health & safety professionals who want the highest-quality training and certification.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation and training products for medical device Quality Management Systems (QMS) according to the ISO 13485 standard.
All required policies, procedures, and forms to implement a medical device QMS according to ISO 13485.
Accredited courses for individuals and medical device professionals who want the highest-quality training and certification.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Compliance products for the European Union’s Medical Device Regulation.
All required policies, procedures, and forms to comply with the EU MDR.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation products for Information Technology Service Management Systems (ITSMS) according to the ISO 20000 standard.
All required policies, procedures, and forms to implement an ITSMS according to ISO 20000.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation products for Business Continuity Management Systems (BCMS) according to the ISO 22301 standard.
All required policies, procedures, and forms to implement a BCMS according to ISO 22301.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation products for testing and calibration laboratories according to the ISO 17025 standard.
All required policies, procedures, and forms to implement ISO 17025 in a laboratory.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation products for automotive Quality Management Systems (QMS) according to the IATF 16949 standard.
All required policies, procedures, and forms to implement an automotive QMS according to IATF 16949.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation products for aerospace Quality Management Systems (QMS) according to the AS9100 standard.
All required policies, procedures, and forms to implement an aerospace QMS according to AS9100.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation, maintenance, training, and knowledge products for consultancies.
Handle multiple ISO 27001 projects by automating repetitive tasks during ISMS implementation.
All required policies, procedures, and forms to implement various standards and regulations for your clients.
Grow your business by organizing cybersecurity and compliance training for your clients under your own brand using Advisera’s learning management system platform.
Accredited Lead Auditor and Implementer courses for DORA and ISO standards, and advanced courses to help consultants grow their business, increase recurring revenue, and stand out from bigger competitors.
Create compliance documents, get instant answers to compliance questions, build training materials faster, and refine writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Find new clients, potential partners, and collaborators and meet a community of like-minded professionals locally and globally.
Implementation, maintenance, training, and knowledge products for the IT industry.
Automate your ISMS implementation and maintenance with the Risk Register, Statement of Applicability, and wizards for all required documents.
Documentation to comply with ISO 27001 (cybersecurity), ISO 22301 (business continuity), ISO 20000 (IT service management), GDPR (privacy), NIS 2 (critical infrastructure cybersecurity), and DORA (cybersecurity for financial sector).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity, privacy, and AI program.
Accredited courses for individuals and security professionals who want the highest-quality training and certification.
Create ISO 27001 and NIS2 documentation, get instant answers to any questions related to ISO 27001 and the ISMS, refine your writing, and build security training materials faster with Advisera’s AI-powered platform
Compliance, training, and knowledge products for essential and important organizations.
Documentation to comply with NIS 2 (cybersecurity), GDPR (privacy), ISO 27001 (cybersecurity), and ISO 22301 (business continuity).
Company-wide training program for employees and senior management to comply with Article 20 of the NIS 2 cybersecurity directive.
Accredited courses for individuals and security professionals who want the highest-quality training and certification.
Create ISO 27001 and NIS2 documentation, get instant answers to any questions related to ISO 27001 (ISMS), refine your writing, and build security awareness training materials faster with Advisera’s AI-powered platform
Implementation, training, and knowledge products for manufacturing companies.
Documentation to comply with ISO 9001 (quality), ISO 14001 (environmental), and ISO 45001 (health & safety), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS) and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for transportation & distribution companies.
Documentation to comply with ISO 9001 (quality), ISO 14001 (environmental), and ISO 45001 (health & safety), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS) and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for schools, universities, and other educational organizations.
Documentation to comply with ISO 27001 (cybersecurity), ISO 9001 (quality), and GDPR (privacy).
Company-wide cybersecurity and AI governance awareness program for all employees, to decrease incidents, support a successful cybersecurity program, and ensure responsible use of AI.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS) and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, maintenance, training, and knowledge products for telecoms.
Automate your ISMS implementation and maintenance with the Risk Register, Statement of Applicability, and wizards for all required documents.
Documentation to comply with ISO 27001 (cybersecurity), ISO 22301 (business continuity), ISO 20000 (IT service management), GDPR (privacy), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity program.
Accredited courses for individuals and security professionals who want the highest-quality training and certification.
Create ISO 27001 and NIS2 documentation, get instant answers to any questions related to ISO 27001 (ISMS), refine your writing, and build security awareness training materials faster with Advisera’s AI-powered platform.
Implementation, maintenance, training, and knowledge products for banks, insurance companies, and other financial organizations.
Automate your ISMS implementation and maintenance with the Risk Register, Statement of Applicability, and wizards for all required documents.
Documentation to comply with DORA (cybersecurity for financial sector), ISO 27001 (cybersecurity), ISO 22301 (business continuity), and GDPR (privacy).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity, privacy, and AI program.
Accredited courses for individuals and security professionals who want the highest-quality training and certification.
Create ISO 27001 and NIS2 documentation, get instant answers to any questions related to ISO 27001 (ISMS), refine your writing, and build security awareness training materials faster with Advisera’s AI-powered platform.
Implementation, training, and knowledge products for local, regional, and national government entities.
Documentation to comply with ISO 27001 (cybersecurity), ISO 9001 (quality), GDPR (privacy), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity, privacy, and AI program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 27001 (ISMS) and ISO 9001 (QMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for hospitals and other health organizations.
Documentation to comply with ISO 27001 (cybersecurity), ISO 9001 (quality), ISO 14001 (environmental), ISO 45001 (health & safety), NIS 2 (critical infrastructure cybersecurity) and GDPR (privacy).
Company-wide cybersecurity and AI governance awareness program for all employees, to decrease incidents, support a successful cybersecurity program, and ensure responsible use of AI.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 27001 (ISMS), ISO 9001 (QMS), and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for the medical device industry.
Documentation to comply with ISO 13485 (medical device), ISO 27001 (cybersecurity), ISO 9001 (quality), ISO 14001 (environmental), ISO 45001 (health & safety), NIS 2 (critical infrastructure cybersecurity) and GDPR (privacy).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity, privacy, and AI program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 27001 (ISMS), ISO 9001 (QMS), and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for the aerospace industry.
Documentation to comply with AS9100 (aerospace), ISO 9001 (quality), ISO 14001 (environmental), and ISO 45001 (health & safety), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS) and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for the automotive industry.
Documentation to comply with IATF 16949 (automotive), ISO 9001 (quality), ISO 14001 (environmental), and ISO 45001 (health & safety), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity and AI program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS) and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for laboratories.
Documentation to comply with ISO 17025 (testing and calibration laboratories), ISO 9001 (quality), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity program.
Accredited courses for individuals and quality professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
 Mark Hammar
Mark Hammar
At some point during your implementation of an ISO 9001 Quality Management System (QMS), you will have to write down some policies and procedures. There is no avoiding this. While it is useful and important to make sure you don’t have too much documentation, there are definitely some policies and procedures that you will need to write down in order to prevent non-conformances. For this reason, you should have a standard way in your organization to write these documents so that they are consistent and easy to understand.
After deciding that a process needs to be documented in a procedure, you first need to know the limits of the process you are documenting. For instance, if you are documenting the purchasing process, it may start with a list of parts to be purchased from the designer, and end when the parts are delivered from the supplier and determined to be acceptable. If this is the case, you will need to detail the requirements for each step that is deemed necessary, such as how purchasing determines who to buy from, how the purchase order is placed, and how the parts are accepted when received. Knowing the limits of the process will help you to know when to stop documenting.
There are different ways to view a process, but making sure you understand the whole process is important to ensure that nothing is missed, even if you don’t need to document everything. One way to view the process is through a turtle diagram, which shows the six different external interactions of a process.
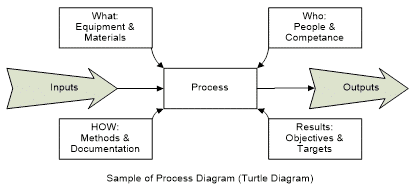
Another method of viewing a process is the SIPOC model often used in Six Sigma improvements. This is an acronym for Supplier, Inputs, Process, Outputs, and Customer. This method makes you focus on how the process you are documenting interacts with other processes before and after it. Whichever ways you choose to look at the process, make sure you look at all aspects of the process when gathering the information for your documentation.
For more information on creating a process flowchart, see this white paper on How to create an ISO 9001 process flowchart.
Aligning with other processes is very important, and making these interactions work well is one of the best ways to make the whole system more efficient. One place to focus your attention is the inputs and outputs of the process. For the inputs, does the process that creates the input know the requirements of the process using it, or might they change the input later? For the process output, is it used somewhere as an input? If not, then you need to question if this output is actually necessary.
There are many different ways to document a procedure. For instance, you may choose to have documentation that is a series of paragraphs, or you may choose to have a procedure that is in a flowchart format. Regardless, many companies have a standard document structure to help people know where to look for certain important pieces of information, such as the applicability of the document. The blog article How to structure quality management system documentation not only shows a structure for documentation hierarchy, but in section 3 also gives a good sample format for a quality management system procedure. Knowing where to look for the information needed can save employees time and improve the efficiency of the QMS documentation.
Take what you have found and write enough detail that competent people reading it can perform the job required without causing non-conformances. This is the reason you have a document. If you add unnecessary information, you not only make your document more cumbersome, but you might confuse your audience. I once saw a procedure that stated: “No eating, drinking or smoking in the lab; effective 24 hours a day 365 days a year.” By adding the last part, does that mean that on February 29th in a leap year you can do this? Think about what you write.
In all cases, you need to make sure the right people approve a document for use. This might be the production manager for production procedures, or the purchasing manager for purchasing procedures. A good QMS will define who need to approve which documents so that the right experts are ensuring that the procedure is correct.
I am not a big proponent of training by having people read procedures and sign a paper as a record that they did. It is best to find some way to make sure that the people who need to use a procedure have not only read it, but also understand and have implemented any changes into the process. This can be done through audits or even questioning employees to make sure they understand the important parts of a procedure. By doing this, you can also check the effectiveness of the documented procedure to see if any improvements need to be made.
It is good to remember that a documented procedure does not necessarily mean that a process is static and unable to change. Through continual review by operators, auditors, and management you can identify potential improvements in your processes and change your documentation accordingly. Sometimes it is hard to see all of this change as a good thing, but if it is driving improvement – it is. Use your documentation to capture improvements so that everyone is using best practices, and you will be farther ahead in your goal of more efficient processes.
To implement ISO 9001 easily and efficiently, use our ISO 9001 Premium Documentation Toolkit that provides step-by-step guidance and all documents for full ISO 9001 compliance.

Mark Hammar is a Certified Manager of Quality / Organizational Excellence through the American Society for Quality and has been a Quality Professional since 1994. Mark has experience in auditing, improving processes, and writing procedures for Quality, Environmental, and Occupational Health & Safety Management Systems, and is certified as a Lead Auditor for ISO 9001, AS9100, and ISO 14001.

You may unsubscribe at any time. For more information, please see our privacy notice.