Strahinja Stojanovic
Strahinja Stojanovic
November 16, 2017
Implementation, maintenance, training, and knowledge products for Information Security Management Systems (ISMS) according to the ISO 27001 standard.
Automate your ISMS implementation and maintenance with the Risk Register, Statement of Applicability, and wizards for all required documents.
All required policies, procedures, and forms to implement an ISMS according to ISO 27001.
Train your key people about ISO 27001 requirements and provide cybersecurity awareness training to all of your employees.
Accredited courses for individuals and security professionals who want the highest-quality training and certification.
Create ISO 27001 documentation, get instant answers to any questions related to ISO 27001 and the ISMS, refine your writing, and build security training materials faster with Advisera’s AI-powered platform.
Compliance and training products for critical infrastructure organizations for the European Union’s Network and Information Systems cybersecurity directive.
All required policies, procedures, and forms to comply with the NIS 2 cybersecurity directive.
Company-wide training program for employees and senior management to comply with Article 20 of the NIS 2 cybersecurity directive.
Create NIS2 documentation, refine your writing, and build security awareness training materials faster with Advisera’s AI-powered platform.
Compliance and training products for financial entities for the European Union’s DORA regulation.
All required policies, procedures, and forms to comply with the DORA regulation.
Company-wide cybersecurity and resilience training program for all employees, to train them and raise awareness about ICT risk management.
Accredited courses for individuals and DORA professionals who want the highest-quality training and certification.
Create DORA documentation, refine your writing, and build financial sector cybersecurity training materials faster with Advisera’s AI-powered platform.
Training products for Artificial Intelligence Management Systems (AIMS) and AI governance according to the ISO 42001 standard.
Accredited courses for individuals, consultants, and AI professionals who want the highest-quality training and certification in AI governance and compliance.
Train your key people on ISO 42001 requirements and provide company-wide AI governance training so employees learn how to use AI responsibly and in compliance with your policies.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Compliance and training products for personal data protection according to the European Union’s General Data Protection Regulation.
All required policies, procedures, and forms to comply with the EU GDPR privacy regulation.
Train your key people about GDPR requirements to ensure awareness of data protection principles, privacy rights, and regulatory compliance.
Accredited courses for individuals and privacy professionals who want the highest-quality training and certification.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation, training, and knowledge products for Quality Management Systems (QMS) according to the ISO 9001 standard.
All required policies, procedures, and forms to implement a QMS according to ISO 9001.
Accredited courses for individuals and quality professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 and the QMS, refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for Environmental Management Systems (EMS) according to the ISO 14001 standard.
All required policies, procedures, and forms to implement an EMS according to ISO 14001.
Accredited courses for individuals and environmental professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 14001 and the EMS, refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation and training products for Occupational Health & Safety Management Systems (OHSMS) according to the ISO 45001 standard.
All required policies, procedures, and forms to implement an OHSMS according to ISO 45001.
Accredited courses for individuals and health & safety professionals who want the highest-quality training and certification.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation and training products for medical device Quality Management Systems (QMS) according to the ISO 13485 standard.
All required policies, procedures, and forms to implement a medical device QMS according to ISO 13485.
Accredited courses for individuals and medical device professionals who want the highest-quality training and certification.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Compliance products for the European Union’s Medical Device Regulation.
All required policies, procedures, and forms to comply with the EU MDR.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation products for Information Technology Service Management Systems (ITSMS) according to the ISO 20000 standard.
All required policies, procedures, and forms to implement an ITSMS according to ISO 20000.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation products for Business Continuity Management Systems (BCMS) according to the ISO 22301 standard.
All required policies, procedures, and forms to implement a BCMS according to ISO 22301.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation products for testing and calibration laboratories according to the ISO 17025 standard.
All required policies, procedures, and forms to implement ISO 17025 in a laboratory.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation products for automotive Quality Management Systems (QMS) according to the IATF 16949 standard.
All required policies, procedures, and forms to implement an automotive QMS according to IATF 16949.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation products for aerospace Quality Management Systems (QMS) according to the AS9100 standard.
All required policies, procedures, and forms to implement an aerospace QMS according to AS9100.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation, maintenance, training, and knowledge products for consultancies.
Handle multiple ISO 27001 projects by automating repetitive tasks during ISMS implementation.
All required policies, procedures, and forms to implement various standards and regulations for your clients.
Grow your business by organizing cybersecurity and compliance training for your clients under your own brand using Advisera’s learning management system platform.
Accredited Lead Auditor and Implementer courses for DORA and ISO standards, and advanced courses to help consultants grow their business, increase recurring revenue, and stand out from bigger competitors.
Create compliance documents, get instant answers to compliance questions, build training materials faster, and refine writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Find new clients, potential partners, and collaborators and meet a community of like-minded professionals locally and globally.
Implementation, maintenance, training, and knowledge products for the IT industry.
Automate your ISMS implementation and maintenance with the Risk Register, Statement of Applicability, and wizards for all required documents.
Documentation to comply with ISO 27001 (cybersecurity), ISO 22301 (business continuity), ISO 20000 (IT service management), GDPR (privacy), NIS 2 (critical infrastructure cybersecurity), and DORA (cybersecurity for financial sector).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity, privacy, and AI program.
Accredited courses for individuals and security professionals who want the highest-quality training and certification.
Create ISO 27001 and NIS2 documentation, get instant answers to any questions related to ISO 27001 and the ISMS, refine your writing, and build security training materials faster with Advisera’s AI-powered platform
Compliance, training, and knowledge products for essential and important organizations.
Documentation to comply with NIS 2 (cybersecurity), GDPR (privacy), ISO 27001 (cybersecurity), and ISO 22301 (business continuity).
Company-wide training program for employees and senior management to comply with Article 20 of the NIS 2 cybersecurity directive.
Accredited courses for individuals and security professionals who want the highest-quality training and certification.
Create ISO 27001 and NIS2 documentation, get instant answers to any questions related to ISO 27001 (ISMS), refine your writing, and build security awareness training materials faster with Advisera’s AI-powered platform
Implementation, training, and knowledge products for manufacturing companies.
Documentation to comply with ISO 9001 (quality), ISO 14001 (environmental), and ISO 45001 (health & safety), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS) and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for transportation & distribution companies.
Documentation to comply with ISO 9001 (quality), ISO 14001 (environmental), and ISO 45001 (health & safety), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS) and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for schools, universities, and other educational organizations.
Documentation to comply with ISO 27001 (cybersecurity), ISO 9001 (quality), and GDPR (privacy).
Company-wide cybersecurity and AI governance awareness program for all employees, to decrease incidents, support a successful cybersecurity program, and ensure responsible use of AI.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS) and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, maintenance, training, and knowledge products for telecoms.
Automate your ISMS implementation and maintenance with the Risk Register, Statement of Applicability, and wizards for all required documents.
Documentation to comply with ISO 27001 (cybersecurity), ISO 22301 (business continuity), ISO 20000 (IT service management), GDPR (privacy), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity program.
Accredited courses for individuals and security professionals who want the highest-quality training and certification.
Create ISO 27001 and NIS2 documentation, get instant answers to any questions related to ISO 27001 (ISMS), refine your writing, and build security awareness training materials faster with Advisera’s AI-powered platform.
Implementation, maintenance, training, and knowledge products for banks, insurance companies, and other financial organizations.
Automate your ISMS implementation and maintenance with the Risk Register, Statement of Applicability, and wizards for all required documents.
Documentation to comply with DORA (cybersecurity for financial sector), ISO 27001 (cybersecurity), ISO 22301 (business continuity), and GDPR (privacy).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity, privacy, and AI program.
Accredited courses for individuals and security professionals who want the highest-quality training and certification.
Create ISO 27001 and NIS2 documentation, get instant answers to any questions related to ISO 27001 (ISMS), refine your writing, and build security awareness training materials faster with Advisera’s AI-powered platform.
Implementation, training, and knowledge products for local, regional, and national government entities.
Documentation to comply with ISO 27001 (cybersecurity), ISO 9001 (quality), GDPR (privacy), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity, privacy, and AI program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 27001 (ISMS) and ISO 9001 (QMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for hospitals and other health organizations.
Documentation to comply with ISO 27001 (cybersecurity), ISO 9001 (quality), ISO 14001 (environmental), ISO 45001 (health & safety), NIS 2 (critical infrastructure cybersecurity) and GDPR (privacy).
Company-wide cybersecurity and AI governance awareness program for all employees, to decrease incidents, support a successful cybersecurity program, and ensure responsible use of AI.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 27001 (ISMS), ISO 9001 (QMS), and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for the medical device industry.
Documentation to comply with ISO 13485 (medical device), ISO 27001 (cybersecurity), ISO 9001 (quality), ISO 14001 (environmental), ISO 45001 (health & safety), NIS 2 (critical infrastructure cybersecurity) and GDPR (privacy).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity, privacy, and AI program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 27001 (ISMS), ISO 9001 (QMS), and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for the aerospace industry.
Documentation to comply with AS9100 (aerospace), ISO 9001 (quality), ISO 14001 (environmental), and ISO 45001 (health & safety), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS) and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for the automotive industry.
Documentation to comply with IATF 16949 (automotive), ISO 9001 (quality), ISO 14001 (environmental), and ISO 45001 (health & safety), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity and AI program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS) and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for laboratories.
Documentation to comply with ISO 17025 (testing and calibration laboratories), ISO 9001 (quality), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity program.
Accredited courses for individuals and quality professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
 Strahinja Stojanovic
Strahinja Stojanovic
Updated: August 29, 2023
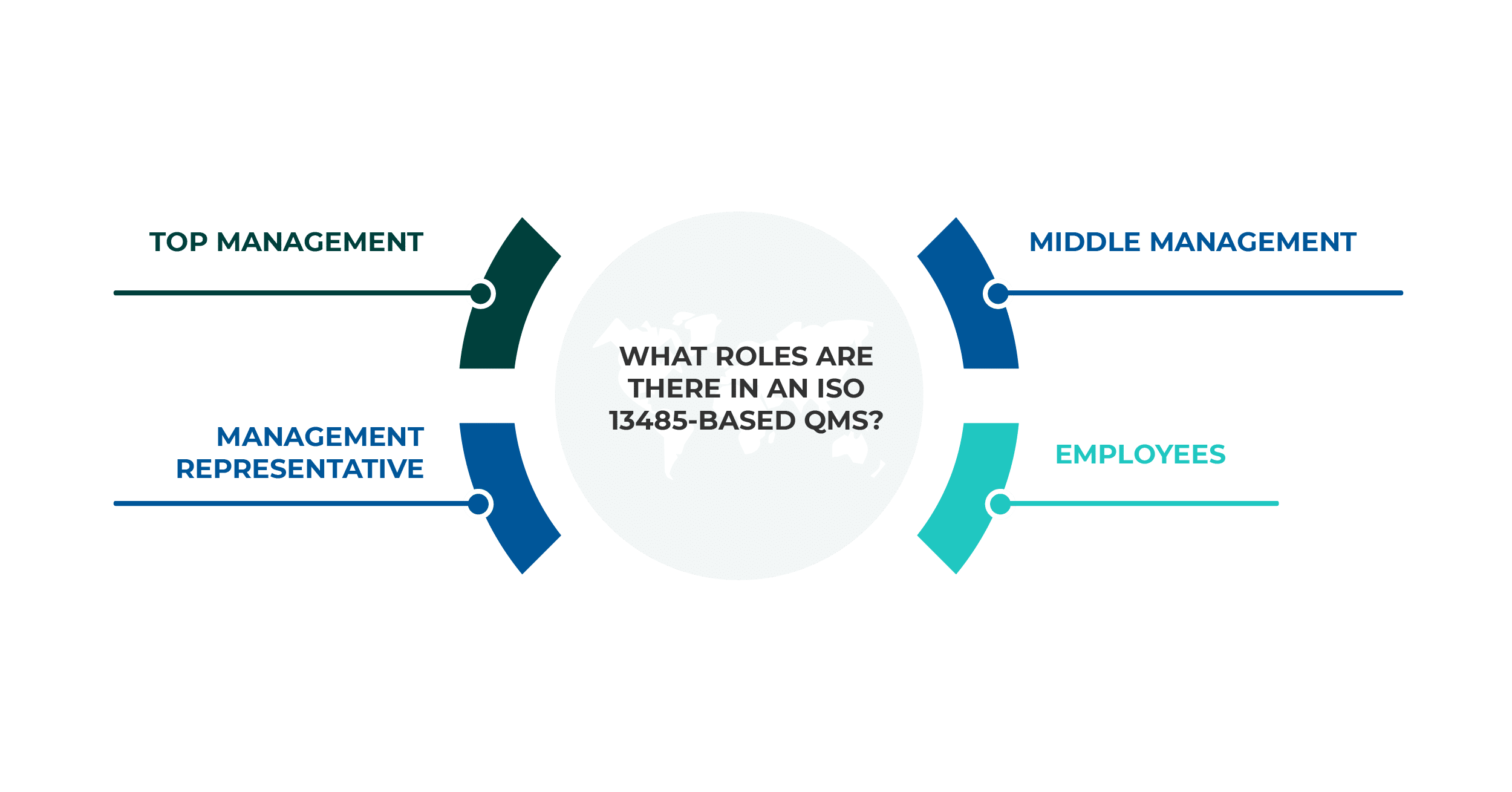
When you are developing a Quality Management System (QMS), you need to assign and document the roles and responsibilities within your management system. ISO 13485 highlights this need, as shown primarily in the requirements of clauses 5.5.1 and 5.5.2. So, what roles and responsibilities do you need to identify, and how should you do this? Here are some ideas about what to do.
The requirements of the standard regarding roles and responsibilities are very loose, so the organization can define them in any way it sees fit. Top management has a responsibility to ensure that ISO 13485 roles and responsibilities within the organization are clearly defined, documented, and communicated. This includes documenting the relationships among personnel who carry out, supervise, and verify work that affects quality, and ensuring that these individuals have the independence and authority necessary to perform their duties effectively.
Implementation failure is often caused by a lack of awareness and commitment to the implementation and maintenance of an ISO 13485-based QMS by the top management. This lack of top management involvement can also mean that the standard isn’t implemented fully. The primary concerns of the top management are to ensure the long-term success of their company, to increase profitability, to control new initiatives, to decrease risks, etc. This means that top management is involved in assessing the effectiveness of the Quality Management System. An increase in top management involvement can be achieved by explaining to them the benefits that ISO 13485 implementation can have for a business, and the potential negative consequences of a poorly established QMS.
Most of the requirements for engagement of the top management in the QMS are stated in clause 5. The first part describes how top management must show leadership through its commitment to the QMS, and the remaining three parts discuss the Quality Policy and its requirements; the need to put customers first; and the roles, responsibilities, and authorities of the company.
To define the appropriate roles, the following processes are necessary:
The responsibilities of top management in ISO 13485 are:
Communicating the importance of meeting customer and regulatory requirements. Like in any other QMS, the focus is on the customer. But, considering how highly regulated the medical device industry is, it is important that top management ensures compliance with these requirements by communicating with the rest of the organization.
Establishing the Quality Policy. Top management needs to establish and publish the Quality Policy, in which they will define the intention of the QMS, and direct everyone in the organization as to how medical devices will be created and delivered to the customers.
Establish the objectives. Through the objectives, top management defines and plans which direction the QMS will follow to meet the requirements outlined in section 4.1. The objectives also provide a clear measure of whether the system is effective.
Find out more here: Setting good quality objectives for ISO 13485.
Conduct the management reviews. Management reviews are the final check to see whether the QMS is effective, and what actions need to be taken for its improvement. The management review process is a key indicator of top management’s ongoing commitment to the Quality Management System.
For more information, see: How to Perform Management Review According to ISO 13485.
Provide all the necessary resources. Without enough money or employee time, the ISO 13485 project will fail; therefore, support from top management must become very real and tangible. From my experience, this is exactly the point where the management usually fails – they usually redirect needed resources into other projects.
When the above requirements are met, top management demonstrates that the QMS is not a side project, but rather an important part of business processes.
As previously mentioned, there must be at least one management representative (MR) who will be in charge of the entire QMS. This person will be the backbone of the system and will have the ultimate responsibility for its effectiveness.
Top management should appoint a member of management who has the responsibility and authority to act as the MR. This individual should be responsible for ensuring that the organization’s Quality Management System is established, implemented, maintained, and continually improved.
The main responsibilities of the ISO 13485 management representative are:
Ensuring the documentation of the processes needed for the QMS – The MR is usually the person who has the most knowledge of the standard in the organization. It is his or her duty to ensure that the documentation complies with the requirements of the standard. For more information, see: List of mandatory documents required by ISO 13485:2016.
Reporting on the performance of the QMS to top management – This includes conducting internal audits, auditing compliance with legal and other requirements, and monitoring the results of the process performance on a regular basis.
Ensuring the promotion of awareness of applicable regulatory and QMS requirements throughout the organization – As mentioned before, compliance with applicable requirements is crucial for the organization, and the MR needs to ensure that the employees are aware of the requirements, as well as the consequences of noncompliance.
All of this sounds like an incredible amount of work – and it is. The MR should delegate these responsibilities to the middle management.
The standard does not specify the required profession or prior knowledge for the position of management representative. While it is assumed that the individual should have knowledge and understanding of quality systems, they are also expected to possess knowledge of the product being produced. Additionally, the ISO 13485 management representative must possess the following skills:
However, it is ultimately up to top management to determine which member of management has the necessary knowledge and abilities to fulfill this role and its associated responsibilities.
Middle management has two significant roles in the QMS. The first is to help with the assessment of risks and the determination of operational controls for activities and processes within their scope. The second role is, of course, to ensure that all rules are followed by the employees.
Because they are the ones enforcing and executing the QMS on a daily basis, their input on how the system works and what should be changed is of utmost value.
Employee engagement depends primarily on how the importance and the purpose of the system are explained to them. Nothing can make the system work (or fail) like the employees’ perception of its importance.
When each employee is clear on their roles and responsibilities, aware of how they contribute to the system, and why it is important for them personally, the organization has an effective QMS. With strong employee engagement, an organization will be able to have an effective QMS and achieve all the benefits that ISO 13485 can bring to the organization.
To implement ISO 13485 easily and efficiently, use our ISO 13485 Documentation Toolkit that provides step-by-step guidance and all documents for full ISO 13485 compliance.

Strahinja Stojanovic is certified as a lead auditor for the ISO 13485, ISO 9001, ISO 14001, and OHSAS 18001 standards by RABQSA. He participated in the implementation of these standards in more than 100 SMEs, through the creation of documentation and performing in-house training for maintaining management systems, internal audits, and management reviews.


You may unsubscribe at any time. For more information, please see our privacy notice.