Kristina Zvonar Brkic
Kristina Zvonar Brkic
March 9, 2021
Implementation, maintenance, training, and knowledge products for Information Security Management Systems (ISMS) according to the ISO 27001 standard.
Automate your ISMS implementation and maintenance with the Risk Register, Statement of Applicability, and wizards for all required documents.
All required policies, procedures, and forms to implement an ISMS according to ISO 27001.
Train your key people about ISO 27001 requirements and provide cybersecurity awareness training to all of your employees.
Accredited courses for individuals and security professionals who want the highest-quality training and certification.
Create ISO 27001 documentation, get instant answers to any questions related to ISO 27001 and the ISMS, refine your writing, and build security training materials faster with Advisera’s AI-powered platform.
Compliance and training products for critical infrastructure organizations for the European Union’s Network and Information Systems cybersecurity directive.
All required policies, procedures, and forms to comply with the NIS 2 cybersecurity directive.
Company-wide training program for employees and senior management to comply with Article 20 of the NIS 2 cybersecurity directive.
Create NIS2 documentation, refine your writing, and build security awareness training materials faster with Advisera’s AI-powered platform.
Compliance and training products for financial entities for the European Union’s DORA regulation.
All required policies, procedures, and forms to comply with the DORA regulation.
Company-wide cybersecurity and resilience training program for all employees, to train them and raise awareness about ICT risk management.
Accredited courses for individuals and DORA professionals who want the highest-quality training and certification.
Create DORA documentation, refine your writing, and build financial sector cybersecurity training materials faster with Advisera’s AI-powered platform.
Training products for Artificial Intelligence Management Systems (AIMS) and AI governance according to the ISO 42001 standard.
Accredited courses for individuals, consultants, and AI professionals who want the highest-quality training and certification in AI governance and compliance.
Train your key people on ISO 42001 requirements and provide company-wide AI governance training so employees learn how to use AI responsibly and in compliance with your policies.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Compliance and training products for personal data protection according to the European Union’s General Data Protection Regulation.
All required policies, procedures, and forms to comply with the EU GDPR privacy regulation.
Train your key people about GDPR requirements to ensure awareness of data protection principles, privacy rights, and regulatory compliance.
Accredited courses for individuals and privacy professionals who want the highest-quality training and certification.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation, training, and knowledge products for Quality Management Systems (QMS) according to the ISO 9001 standard.
All required policies, procedures, and forms to implement a QMS according to ISO 9001.
Accredited courses for individuals and quality professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 and the QMS, refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for Environmental Management Systems (EMS) according to the ISO 14001 standard.
All required policies, procedures, and forms to implement an EMS according to ISO 14001.
Accredited courses for individuals and environmental professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 14001 and the EMS, refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation and training products for Occupational Health & Safety Management Systems (OHSMS) according to the ISO 45001 standard.
All required policies, procedures, and forms to implement an OHSMS according to ISO 45001.
Accredited courses for individuals and health & safety professionals who want the highest-quality training and certification.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation and training products for medical device Quality Management Systems (QMS) according to the ISO 13485 standard.
All required policies, procedures, and forms to implement a medical device QMS according to ISO 13485.
Accredited courses for individuals and medical device professionals who want the highest-quality training and certification.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Compliance products for the European Union’s Medical Device Regulation.
All required policies, procedures, and forms to comply with the EU MDR.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation products for Information Technology Service Management Systems (ITSMS) according to the ISO 20000 standard.
All required policies, procedures, and forms to implement an ITSMS according to ISO 20000.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation products for Business Continuity Management Systems (BCMS) according to the ISO 22301 standard.
All required policies, procedures, and forms to implement a BCMS according to ISO 22301.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation products for testing and calibration laboratories according to the ISO 17025 standard.
All required policies, procedures, and forms to implement ISO 17025 in a laboratory.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation products for automotive Quality Management Systems (QMS) according to the IATF 16949 standard.
All required policies, procedures, and forms to implement an automotive QMS according to IATF 16949.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation products for aerospace Quality Management Systems (QMS) according to the AS9100 standard.
All required policies, procedures, and forms to implement an aerospace QMS according to AS9100.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation, maintenance, training, and knowledge products for consultancies.
Handle multiple ISO 27001 projects by automating repetitive tasks during ISMS implementation.
All required policies, procedures, and forms to implement various standards and regulations for your clients.
Grow your business by organizing cybersecurity and compliance training for your clients under your own brand using Advisera’s learning management system platform.
Accredited Lead Auditor and Implementer courses for DORA and ISO standards, and advanced courses to help consultants grow their business, increase recurring revenue, and stand out from bigger competitors.
Create compliance documents, get instant answers to compliance questions, build training materials faster, and refine writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Find new clients, potential partners, and collaborators and meet a community of like-minded professionals locally and globally.
Implementation, maintenance, training, and knowledge products for the IT industry.
Automate your ISMS implementation and maintenance with the Risk Register, Statement of Applicability, and wizards for all required documents.
Documentation to comply with ISO 27001 (cybersecurity), ISO 22301 (business continuity), ISO 20000 (IT service management), GDPR (privacy), NIS 2 (critical infrastructure cybersecurity), and DORA (cybersecurity for financial sector).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity, privacy, and AI program.
Accredited courses for individuals and security professionals who want the highest-quality training and certification.
Create ISO 27001 and NIS2 documentation, get instant answers to any questions related to ISO 27001 and the ISMS, refine your writing, and build security training materials faster with Advisera’s AI-powered platform
Compliance, training, and knowledge products for essential and important organizations.
Documentation to comply with NIS 2 (cybersecurity), GDPR (privacy), ISO 27001 (cybersecurity), and ISO 22301 (business continuity).
Company-wide training program for employees and senior management to comply with Article 20 of the NIS 2 cybersecurity directive.
Accredited courses for individuals and security professionals who want the highest-quality training and certification.
Create ISO 27001 and NIS2 documentation, get instant answers to any questions related to ISO 27001 (ISMS), refine your writing, and build security awareness training materials faster with Advisera’s AI-powered platform
Implementation, training, and knowledge products for manufacturing companies.
Documentation to comply with ISO 9001 (quality), ISO 14001 (environmental), and ISO 45001 (health & safety), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS) and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for transportation & distribution companies.
Documentation to comply with ISO 9001 (quality), ISO 14001 (environmental), and ISO 45001 (health & safety), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS) and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for schools, universities, and other educational organizations.
Documentation to comply with ISO 27001 (cybersecurity), ISO 9001 (quality), and GDPR (privacy).
Company-wide cybersecurity and AI governance awareness program for all employees, to decrease incidents, support a successful cybersecurity program, and ensure responsible use of AI.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS) and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, maintenance, training, and knowledge products for telecoms.
Automate your ISMS implementation and maintenance with the Risk Register, Statement of Applicability, and wizards for all required documents.
Documentation to comply with ISO 27001 (cybersecurity), ISO 22301 (business continuity), ISO 20000 (IT service management), GDPR (privacy), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity program.
Accredited courses for individuals and security professionals who want the highest-quality training and certification.
Create ISO 27001 and NIS2 documentation, get instant answers to any questions related to ISO 27001 (ISMS), refine your writing, and build security awareness training materials faster with Advisera’s AI-powered platform.
Implementation, maintenance, training, and knowledge products for banks, insurance companies, and other financial organizations.
Automate your ISMS implementation and maintenance with the Risk Register, Statement of Applicability, and wizards for all required documents.
Documentation to comply with DORA (cybersecurity for financial sector), ISO 27001 (cybersecurity), ISO 22301 (business continuity), and GDPR (privacy).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity, privacy, and AI program.
Accredited courses for individuals and security professionals who want the highest-quality training and certification.
Create ISO 27001 and NIS2 documentation, get instant answers to any questions related to ISO 27001 (ISMS), refine your writing, and build security awareness training materials faster with Advisera’s AI-powered platform.
Implementation, training, and knowledge products for local, regional, and national government entities.
Documentation to comply with ISO 27001 (cybersecurity), ISO 9001 (quality), GDPR (privacy), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity, privacy, and AI program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 27001 (ISMS) and ISO 9001 (QMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for hospitals and other health organizations.
Documentation to comply with ISO 27001 (cybersecurity), ISO 9001 (quality), ISO 14001 (environmental), ISO 45001 (health & safety), NIS 2 (critical infrastructure cybersecurity) and GDPR (privacy).
Company-wide cybersecurity and AI governance awareness program for all employees, to decrease incidents, support a successful cybersecurity program, and ensure responsible use of AI.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 27001 (ISMS), ISO 9001 (QMS), and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for the medical device industry.
Documentation to comply with ISO 13485 (medical device), ISO 27001 (cybersecurity), ISO 9001 (quality), ISO 14001 (environmental), ISO 45001 (health & safety), NIS 2 (critical infrastructure cybersecurity) and GDPR (privacy).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity, privacy, and AI program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 27001 (ISMS), ISO 9001 (QMS), and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for the aerospace industry.
Documentation to comply with AS9100 (aerospace), ISO 9001 (quality), ISO 14001 (environmental), and ISO 45001 (health & safety), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS) and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for the automotive industry.
Documentation to comply with IATF 16949 (automotive), ISO 9001 (quality), ISO 14001 (environmental), and ISO 45001 (health & safety), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity and AI program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS) and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for laboratories.
Documentation to comply with ISO 17025 (testing and calibration laboratories), ISO 9001 (quality), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity program.
Accredited courses for individuals and quality professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
 Kristina Zvonar Brkic
Kristina Zvonar Brkic
Updated: October 19, 2023.
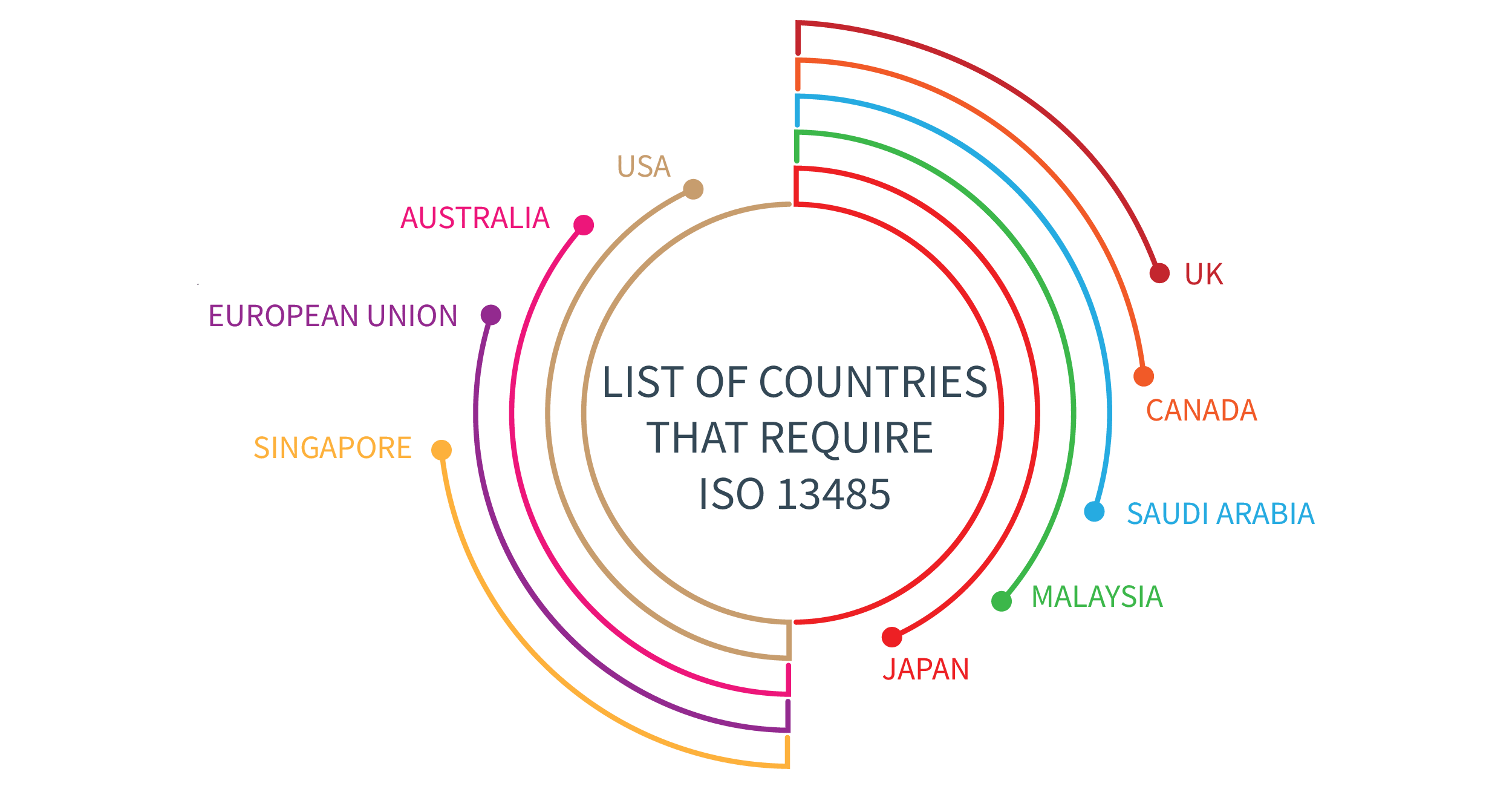
Regulatory requirements for medical devices are becoming tighter every year. Manufacturers are expected to prove that their quality management processes are under control to ensure best practice in everything they do, while observing an increasing number of regulations in many countries. ISO 13485 has gained significant recognition around the world; but, depending on the country, there are other regulations that manufacturers must comply with. Is ISO 13485 an international standard? Is ISO 13485 required in the USA, Canada, and Japan? Is it mandatory in Europe? Where is ISO 13485 required? Learn in this article.
The international standard ISO 13485 is intended to help organizations that are involved in the design, production, installation, and servicing of medical devices and related services to build Quality Management Systems (QMS). A QMS that conforms to ISO 13485 requirements is a set of documented and interrelated processes, including any necessary forms or templates, which are established, implemented, and maintained to fulfill the provisions outlined in the standard’s requirements. The aim of the QMS is to meet the customer and regulatory requirements for businesses operating in the medical device sector. But ISO 13485 is not the only standard that medical device manufacturers must be in compliance with. Each of the listed markets has its own regulation for medical products, with which manufacturers must also comply, in addition to ISO 13485:
| EU | MDR 2017/745 |
| UK | UK MDR 2002 |
| USA | FDA Title 21 CFR Part 807 |
| Canada | Canadian Medical Device Regulations (SOR 98-282) |
| Saudi Arabia | Medical device and supplies regulation |
| Japan | Application for Accreditation |
| Malaysia | Medical device regulation 2012 |
| Singapore | Health Products (Medical Device) Regulations 2010 |
| Australia | Therapeutic goods (medical devices) regulations 2002 |
Now, let’s see more about which countries and regions are required to certify against ISO 13485
Europe. Companies active in the EU market are expected to have a Quality Management System (QMS) certified under ISO 13485:2016, according to the MDR 2017/745. This applies to the following business entities:
UK. With Brexit, the UK has prepared their own set of rules for registering medical devices on the UK market under the Medical Device Regulation 2002 (SI 2002 No 618, as amended) (UK MDR 2002). ISO 13485:2016 is recognized as the designated standard for the UK MDR 2002.
Canada. The Canadian Medical Device Regulations (SOR 98-282) require QMS certification under the Canadian version CAN/CSA-ISO 13485:2016, Medical devices — Quality management systems — Requirements for regulatory purposes.
USA. The US Food & Drug Administration (FDA) has Title 21 Code of Federal Regulation (CFR) Part 820 Quality System Regulation. This regulation is the current quality system for medical devices used by the FDA. While there are many similarities, ISO 13485:2016 is more up to date than 21 CFR 820. But because ISO 13485 is so widespread, the FDA issued in February 2022 a proposed rule to harmonize the US Quality System Regulation (21 CFR 820) with ISO 13485 and make ISO 13485 mandatory. The final rule will replace the current QSR, and the new rule will be referred to as the Quality Management System Regulation (“QMSR”). The FDA proposes to make the QMSR effective one year after the publication date of the final QMSR rule in the Federal Register.
Saudi Arabia. In the document Requirements for Inspections and Quality Management System for Medical Devices, is stated that Quality Management System for medical device manufacturers must be according to the latest version of the Saudi Standard (SFDA.MD/GSO ISO 13485) or its equivalent.
Japan. Japan has largely embraced ISO 13485 as the basis for their QMS requirements. However, Japan’s Ministerial Ordinance on Standards for Manufacturing Control and Quality Control for Medical Devices and In-Vitro Diagnostics (MHLW MO 169) contains additional QMS requirements that medical device manufacturers must meet to be in full compliance. MHLW MO 169 was revised to align with ISO 13485:2016 on March 26, 2021. The transition period is three years, no later than March 25, 2024. Correspondence between ISO 13485:2016 and MHLW MO 169 is on the following link: https://www.pmda.go.jp/files/000239873.pdf.
Malaysia. The Medical device act (Act 737) requires all manufacturers to have implemented a Quality Management System according to the ISO 13485 standard. Other entities, like distributors, importers, and authorized representatives, must be in compliance with the good distribution practice of medical devices (GDPMD).
Singapore. Singapore requires that medical device manufacturers have an ISO 13485-compliant QMS as described in the document Health Products (Medical Device) Regulations 2010.
Australia. The document Therapeutic Goods (Conformity Assessment Standard for Quality Management Systems) Order 2019, published on March 26, 2019, states that the conformity assessment standard for Quality Management Systems is ISO 13485:2016.
Globally recognized certification
From this review, it can be seen that ISO 13485 is recognized worldwide as a major standard in quality assurance systems for medical device manufacturers. Although some markets have some updates, ISO 13485 still remains the main base.
To comply with all ISO 13485 requirements, use this helpful ISO 13485 Documentation Toolkit that provides QMS documents for medical device companies.


You may unsubscribe at any time. For more information, please see our privacy notice.