Kristina Zvonar Brkic
Kristina Zvonar Brkic
March 29, 2021
Implementation, maintenance, training, and knowledge products for Information Security Management Systems (ISMS) according to the ISO 27001 standard.
Automate your ISMS implementation and maintenance with the Risk Register, Statement of Applicability, and wizards for all required documents.
All required policies, procedures, and forms to implement an ISMS according to ISO 27001.
Train your key people about ISO 27001 requirements and provide cybersecurity awareness training to all of your employees.
Accredited courses for individuals and security professionals who want the highest-quality training and certification.
Create ISO 27001 documentation, get instant answers to any questions related to ISO 27001 and the ISMS, refine your writing, and build security training materials faster with Advisera’s AI-powered platform.
Compliance and training products for critical infrastructure organizations for the European Union’s Network and Information Systems cybersecurity directive.
All required policies, procedures, and forms to comply with the NIS 2 cybersecurity directive.
Company-wide training program for employees and senior management to comply with Article 20 of the NIS 2 cybersecurity directive.
Create NIS2 documentation, refine your writing, and build security awareness training materials faster with Advisera’s AI-powered platform.
Compliance and training products for financial entities for the European Union’s DORA regulation.
All required policies, procedures, and forms to comply with the DORA regulation.
Company-wide cybersecurity and resilience training program for all employees, to train them and raise awareness about ICT risk management.
Accredited courses for individuals and DORA professionals who want the highest-quality training and certification.
Create DORA documentation, refine your writing, and build financial sector cybersecurity training materials faster with Advisera’s AI-powered platform.
Training products for Artificial Intelligence Management Systems (AIMS) and AI governance according to the ISO 42001 standard.
Accredited courses for individuals, consultants, and AI professionals who want the highest-quality training and certification in AI governance and compliance.
Train your key people on ISO 42001 requirements and provide company-wide AI governance training so employees learn how to use AI responsibly and in compliance with your policies.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Compliance and training products for personal data protection according to the European Union’s General Data Protection Regulation.
All required policies, procedures, and forms to comply with the EU GDPR privacy regulation.
Train your key people about GDPR requirements to ensure awareness of data protection principles, privacy rights, and regulatory compliance.
Accredited courses for individuals and privacy professionals who want the highest-quality training and certification.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation, training, and knowledge products for Quality Management Systems (QMS) according to the ISO 9001 standard.
All required policies, procedures, and forms to implement a QMS according to ISO 9001.
Accredited courses for individuals and quality professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 and the QMS, refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for Environmental Management Systems (EMS) according to the ISO 14001 standard.
All required policies, procedures, and forms to implement an EMS according to ISO 14001.
Accredited courses for individuals and environmental professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 14001 and the EMS, refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation and training products for Occupational Health & Safety Management Systems (OHSMS) according to the ISO 45001 standard.
All required policies, procedures, and forms to implement an OHSMS according to ISO 45001.
Accredited courses for individuals and health & safety professionals who want the highest-quality training and certification.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation and training products for medical device Quality Management Systems (QMS) according to the ISO 13485 standard.
All required policies, procedures, and forms to implement a medical device QMS according to ISO 13485.
Accredited courses for individuals and medical device professionals who want the highest-quality training and certification.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Compliance products for the European Union’s Medical Device Regulation.
All required policies, procedures, and forms to comply with the EU MDR.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation products for Information Technology Service Management Systems (ITSMS) according to the ISO 20000 standard.
All required policies, procedures, and forms to implement an ITSMS according to ISO 20000.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation products for Business Continuity Management Systems (BCMS) according to the ISO 22301 standard.
All required policies, procedures, and forms to implement a BCMS according to ISO 22301.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation products for testing and calibration laboratories according to the ISO 17025 standard.
All required policies, procedures, and forms to implement ISO 17025 in a laboratory.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation products for automotive Quality Management Systems (QMS) according to the IATF 16949 standard.
All required policies, procedures, and forms to implement an automotive QMS according to IATF 16949.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation products for aerospace Quality Management Systems (QMS) according to the AS9100 standard.
All required policies, procedures, and forms to implement an aerospace QMS according to AS9100.
Build structured training materials faster and refine your writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Implementation, maintenance, training, and knowledge products for consultancies.
Handle multiple ISO 27001 projects by automating repetitive tasks during ISMS implementation.
All required policies, procedures, and forms to implement various standards and regulations for your clients.
Grow your business by organizing cybersecurity and compliance training for your clients under your own brand using Advisera’s learning management system platform.
Accredited Lead Auditor and Implementer courses for DORA and ISO standards, and advanced courses to help consultants grow their business, increase recurring revenue, and stand out from bigger competitors.
Create compliance documents, get instant answers to compliance questions, build training materials faster, and refine writing using Advisera’s AI-powered platform built on proprietary compliance knowledge.
Find new clients, potential partners, and collaborators and meet a community of like-minded professionals locally and globally.
Implementation, maintenance, training, and knowledge products for the IT industry.
Automate your ISMS implementation and maintenance with the Risk Register, Statement of Applicability, and wizards for all required documents.
Documentation to comply with ISO 27001 (cybersecurity), ISO 22301 (business continuity), ISO 20000 (IT service management), GDPR (privacy), NIS 2 (critical infrastructure cybersecurity), and DORA (cybersecurity for financial sector).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity, privacy, and AI program.
Accredited courses for individuals and security professionals who want the highest-quality training and certification.
Create ISO 27001 and NIS2 documentation, get instant answers to any questions related to ISO 27001 and the ISMS, refine your writing, and build security training materials faster with Advisera’s AI-powered platform
Compliance, training, and knowledge products for essential and important organizations.
Documentation to comply with NIS 2 (cybersecurity), GDPR (privacy), ISO 27001 (cybersecurity), and ISO 22301 (business continuity).
Company-wide training program for employees and senior management to comply with Article 20 of the NIS 2 cybersecurity directive.
Accredited courses for individuals and security professionals who want the highest-quality training and certification.
Create ISO 27001 and NIS2 documentation, get instant answers to any questions related to ISO 27001 (ISMS), refine your writing, and build security awareness training materials faster with Advisera’s AI-powered platform
Implementation, training, and knowledge products for manufacturing companies.
Documentation to comply with ISO 9001 (quality), ISO 14001 (environmental), and ISO 45001 (health & safety), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS) and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for transportation & distribution companies.
Documentation to comply with ISO 9001 (quality), ISO 14001 (environmental), and ISO 45001 (health & safety), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS) and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for schools, universities, and other educational organizations.
Documentation to comply with ISO 27001 (cybersecurity), ISO 9001 (quality), and GDPR (privacy).
Company-wide cybersecurity and AI governance awareness program for all employees, to decrease incidents, support a successful cybersecurity program, and ensure responsible use of AI.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS) and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, maintenance, training, and knowledge products for telecoms.
Automate your ISMS implementation and maintenance with the Risk Register, Statement of Applicability, and wizards for all required documents.
Documentation to comply with ISO 27001 (cybersecurity), ISO 22301 (business continuity), ISO 20000 (IT service management), GDPR (privacy), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity program.
Accredited courses for individuals and security professionals who want the highest-quality training and certification.
Create ISO 27001 and NIS2 documentation, get instant answers to any questions related to ISO 27001 (ISMS), refine your writing, and build security awareness training materials faster with Advisera’s AI-powered platform.
Implementation, maintenance, training, and knowledge products for banks, insurance companies, and other financial organizations.
Automate your ISMS implementation and maintenance with the Risk Register, Statement of Applicability, and wizards for all required documents.
Documentation to comply with DORA (cybersecurity for financial sector), ISO 27001 (cybersecurity), ISO 22301 (business continuity), and GDPR (privacy).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity, privacy, and AI program.
Accredited courses for individuals and security professionals who want the highest-quality training and certification.
Create ISO 27001 and NIS2 documentation, get instant answers to any questions related to ISO 27001 (ISMS), refine your writing, and build security awareness training materials faster with Advisera’s AI-powered platform.
Implementation, training, and knowledge products for local, regional, and national government entities.
Documentation to comply with ISO 27001 (cybersecurity), ISO 9001 (quality), GDPR (privacy), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity, privacy, and AI program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 27001 (ISMS) and ISO 9001 (QMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for hospitals and other health organizations.
Documentation to comply with ISO 27001 (cybersecurity), ISO 9001 (quality), ISO 14001 (environmental), ISO 45001 (health & safety), NIS 2 (critical infrastructure cybersecurity) and GDPR (privacy).
Company-wide cybersecurity and AI governance awareness program for all employees, to decrease incidents, support a successful cybersecurity program, and ensure responsible use of AI.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 27001 (ISMS), ISO 9001 (QMS), and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for the medical device industry.
Documentation to comply with ISO 13485 (medical device), ISO 27001 (cybersecurity), ISO 9001 (quality), ISO 14001 (environmental), ISO 45001 (health & safety), NIS 2 (critical infrastructure cybersecurity) and GDPR (privacy).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity, privacy, and AI program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 27001 (ISMS), ISO 9001 (QMS), and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for the aerospace industry.
Documentation to comply with AS9100 (aerospace), ISO 9001 (quality), ISO 14001 (environmental), and ISO 45001 (health & safety), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS) and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for the automotive industry.
Documentation to comply with IATF 16949 (automotive), ISO 9001 (quality), ISO 14001 (environmental), and ISO 45001 (health & safety), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity and AI program.
Accredited courses for individuals and professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS) and ISO 14001 (EMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
Implementation, training, and knowledge products for laboratories.
Documentation to comply with ISO 17025 (testing and calibration laboratories), ISO 9001 (quality), and NIS 2 (critical infrastructure cybersecurity).
Company-wide cybersecurity awareness program for all employees, to decrease incidents and support a successful cybersecurity program.
Accredited courses for individuals and quality professionals who want the highest-quality training and certification.
Get instant answers to any questions related to ISO 9001 (QMS), refine your writing, and build training materials faster with Advisera’s AI-powered platform built on proprietary compliance expertise.
 Kristina Zvonar Brkic
Kristina Zvonar Brkic
In the ISO 13485:2016 Quality Management System (QMS), management of suppliers is a critical part of the success of the purchasing process. So, what do you need to do for supplier management, and how can a supplier audit help with this part of the purchasing process?
One of the purchasing controls to ensure that you get the right products for your medical device manufacturing is supplier evaluation. Your supplier evaluation, and subsequent supplier monitoring, are intended to be based on the risks that each supplier poses to your ability to provide medical devices that meet customer and regulatory requirements.
Supplier evaluation is not intended to be a one-time thing: once a supplier is approved for use, monitoring of performance is crucial to ensure that they continue to meet your requirements. Failure to meet requirements needs to be addressed and corrected as soon as it is noticed. This approval, monitoring, and any necessary correction of suppliers needs to be demonstrated by records that are maintained to demonstrate that the suppliers are capable of meeting requirements.
While not strictly a requirement of the standard, many companies maintain a list of the suppliers who are currently approved for use by the purchasing department. This list, which includes the dates for re-evaluations, ensures that you always use a supplier who is in good standing with your company.
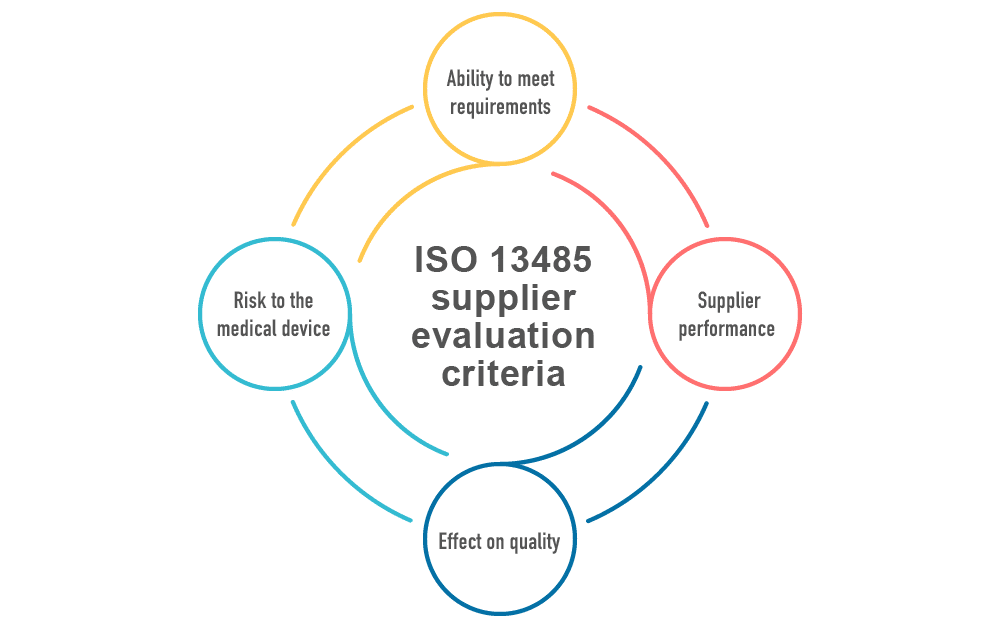
So, how do you make sure that suppliers will meet your needs? ISO 13485 contains a list of things to consider when approving a supplier. These include:
Ability to meet requirements – How well does the supplier meet your requirements? Can they meet the tolerances you need? Are they compliant with any legal requirements that are necessary to provide their products? For example, if there is a certificate of quality or certificate of analysis for purchased material, are the tolerances listed on the certificate in accordance with your requirements?
Supplier performance – Along with technical requirements, how well does the supplier meet your needs? Are they delivering on time? Do they reply in a timely manner to your requests? Can you be sure this supply partner will get you what you need? For example, does the supplier have the appropriate and valid quality certificates (e.g., ISO 9001, ISO 14001, and ISO 13485)?
Effect on quality – How much of an effect does the supplied product have on your medical device manufacturing? If there is a great effect, and the product is highly influential on the quality of your medical device, then more care should be taken in choosing the right supplier. If there is little effect, such as when you have many suppliers for one type of part, then a less rigid approach might be considered.
Risk to the medical device – Along with quality, how does the supplied product affect the safety of the medical device? If it is critical for safe function and operation, then once again, this should increase the level of care in approving the supplier for use.
For more on the overall purchasing process in ISO 13485, see the article: How can ISO 13485 clause 7.4, Purchasing, enhance procurement?
There can be many ways to evaluate if a supplier will meet your needs, or continue to fulfill your requirements. This may include having the supplier prove their capacity and quality using a questionnaire, a survey, or even a trial purchase order, which would allow you to check what they can supply and how good the product is. Another common tool used for approving a medical device supplier is a supplier audit.
Supplier audits, while not an ISO 13485 requirement, provide you with a view into how the processes of the supplier work. Like all management system audits, the supplier audit (also called a second-party audit) is intended to review the processes of the supplier by comparing what is actually happening in the processes against the planned arrangements of the processes—or, in other words, the requirements of the process.
The audit first reviews the process requirements to determine what should be happening, and then collects objective evidence of what is actually happening through observation, conducting interviews, and reviewing records. When this objective evidence is compared to the requirements, the audit can determine if the requirements are being met, which is called process conformity, or if the requirements are not being met, which is called process nonconformity. When a nonconformity exists in a process, then corrective action is needed to fix what is wrong.
Remember that the requirements you have placed on the supplier should be included in those checked during the audit. This is where the supplier audit differs from other management system audits. Instead of reviewing all processes of the supplier against ISO 13485, which is already done by the certification body if they have a certificate, you should focus on the processes that are critical to ensuring that the supplier meets your needs, such as how they deal with nonconforming products, how they ensure they meet legal requirements, and how they review your contracts to ensure that all requirements are met. The processes that are critical to your success are the processes you want to review in the supplier audit.
For example, ensuring traceability is very important in medical device manufacturing. Therefore, it is good to check during the supplier audit how the supplier has ensured traceability from the entry of raw materials to the finished product. Another example is ensuring cleanliness when it matters. During the supplier audit, it can be checked how the cleaning of the production part is prescribed, with which detergents, and how the cleaning process itself is validated.
Along with limiting the scope of your supplier audits to those processes critical to you, it is also beneficial to have your audits focus on how to identify supplier improvements that can help them better meet your needs. This focus on opportunities for improvement, along with a collaborative approach to the audit, will not only help make the audit more beneficial to the supplier, but also help them to better meet your needs.
If the supplier also sees benefit in your approach, it will make the audit much more effective and efficient both for you and the supplier. It is in your best interest to make the supplier audit a win-win scenario.
For a better understanding of the audit process, see this free online training: ISO 13485 Internal Auditor Course.


You may unsubscribe at any time. For more information, please see our privacy notice.